Sugar needs no introduction. Among the first things many of us do in the morning is pour sugar into a cup of coffee. In fact, the sweet substance has become so ubiquitous to modern life that many people must actively monitor their intake so as not to ingest too much. This isn’t surprising news to anyone, yet our sugar consumption habits have not always been this way. While humans have been naturally drawn to sugar since the dawn of mankind, this abundance of sugar is a relatively new phenomenon.
In this Union Kitchen guide, we will examine the science and the history of sugar. This will give you a new appreciation for the substance that has quite literally changed the course of human history.
Composition of Sugar
Despite the vast quantities we consume, most of us know little about sugar beyond its iconic sweet taste. Let’s get molecular and explore one of our favorite substances!
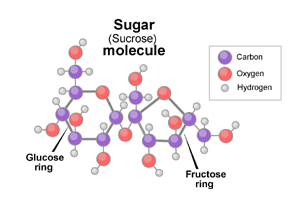
The most common form of sugar that we consume is a simple carbohydrate known as sucrose. Sucrose itself is a combination of two even simpler forms of sugars known as fructose and glucose. Despite this, sugar’s molecular structure (see below) is actually quite complex! It consists of 12 carbon atoms, 22 hydrogen atoms, and 11 oxygen atoms. Most plants naturally produce sugar at some level, though some produce more than others.
What does this all mean for us? Well, most herbivorous and omnivorous animals, including humans, are drawn to sugar thanks to the high amounts of energy it contains in its complex molecular structure.
Let’s go back to those two simple sugars: Glucose and Fructose.
Glucose is what gives us our favorite jolt and source of energy. That’s why people who eat a large sum of sugar (beyond anything a human is supposed to consume) experience something referred to as a sugar high.
Fructose, on the other hand, helps humans store fat, vital in times of want. While the threat of starvation is more foreign to us now, it was a very real danger for most of human history. This explains our cravings for everything sugar, including in that morning cup of joe.
Applications in Cooking
Besides making everything taste sweeter, sugar serves several vital roles in the baking process of many of our favorite recipes.
Sugar holds in moisture, which makes it a natural preserver. As discussed in a previous guide on Dehydration, moisture enables microscopic bacteria to thrive in food, which causes it to spoil more quickly. Since sugar holds in the moisture, the bacteria doesn’t have the necessary environment to thrive. This enables desserts like cake and cookies to stay good for longer. The technique of using sugar for preservation is known as sugaring.
Bio-History of Sugar
When a band of nomads, wandering the east African plains 200,000 years ago, came across sugary food, it was important to take advantage of this energy-rich opportunity.
How would they know which foods were rich in sugar? The same way we do today. They would taste it. Your tongue is incredibly sensitive and is able to pick up on many different tastes. Sweetness is a clear sign that a food contains sugar. This helped our ancestors distinguish the sweet, high-energy foods, like berries and honey, from the potentially-poisonous bitter and sour foods. If you want to step into the shoes (or lack thereof) of our sugar-seeking ancestors, grab a wild plant guide and head into a forest.

Today, sugar plays a massive role in our diet. Thanks to industrialization, sugar transformed from an occasional treat to an everyday staple. It is present in many of the foods we eat, from ice cream to cereal. Basic dessert recipes often call for sugar amounts that would have been unavailable to all but the wealthiest several centuries ago. Those of us who love our desserts should certainly consider ourselves extremely lucky to live in a time where sugar is so readily available. Of course with the good comes the bad. This wide-spread availability has led to an uptick in recent years of many preventable diseases such as: obesity, type 2 diabetes, and heart disease. That’s not to say that you shouldn’t enjoy sugar. Sugar, like everything else, is fine and often wonderfully delectable to eat in moderation.
Needless to say, a lot has changed since the days of foraging for berries. Hopefully, you’ve found this brief overview of the science and history of sugar interesting. For more guides like this, checkout Union Kitchen’s resource guide.